argenx (Euronext & Nasdaq: ARGX), a global immunology company committed to improving the lives of people suffering from severe autoimmune diseases, today reported financial results for the full year 2022 and provided a fourth quarter business update.
In a separate press release, argenx also announced today the appointment of Karen Massey to Chief Operating Officer as part of a planned transition. Ms. Massey will succeed Keith Woods who will remain at the Company through the launch of subcutaneous (SC) efgartigimod after which he will retire and serve as a strategic advisor on the argenx Board of Directors.
“We began 2023 from a position of strength following the successful first year of our VYVGART launch where we were able to reach more than 3,000 gMG patients globally with our transformative therapy. Our focus for the year ahead is expansion; both through upcoming regulatory approvals and launches, and more broadly by reaching additional patient segments with the planned launch of SC efgartigimod and through our ongoing stakeholder engagement efforts,” said Tim Van Hauwermeiren, Chief Executive Officer of argenx. “Looking ahead, we have the opportunity this year to showcase the innovation within our pipeline through multiple data catalysts, including three pivotal data readouts from efgartigimod and the first clinical efficacy data from ARGX-117, our next pipeline-in-a-product candidate. We are well-equipped to build on this significant momentum as we advance our mission to redefine the treatment of autoimmune disease.”
FOURTH QUARTER 2022 AND RECENT BUSINESS UPDATE
VYVGART Expansion
VYVGART is the first-and-only approved neonatal Fc receptor (FcRn) blocker in the U.S., Japan and the EU. argenx is planning for multi-dimensional expansion to reach more patients with VYVGART through additional regulatory approvals for generalized myasthenia gravis (gMG), the launch of SC efgartigimod for gMG, and new autoimmune indications with the VYVGART regulatory submission for immune thrombocytopenia (ITP) in Japan.
- Generated global net VYVGART revenues of $173 million in the fourth quarter of 2022 and $401 million in the full year of 2022
- Additional VYVGART regulatory approvals and commercial launches expected in 2023
- Approval decision expected in Canada in third quarter of 2023 and in China and Israel by end of 2023
- gMG launch in France, United Kingdom and Italy expected by end of 2023 following review of respective reimbursement dossiers
- Regulatory reviews of SC efgartigimod for gMG ongoing in the U.S., EU and Japan
- Prescription Drug User Fee Act (PDUFA) target action date extended to June 20, 2023 following notification from the U.S. Food and Drug Administration (FDA) in January 2023
- Marketing authorization application (MAA) filed in Japan in first quarter of 2023 with approval decision expected by first quarter of 2024
- MAA review underway by European Medicines Agency with approval decision expected in fourth quarter of 2023
- Submission of MAA in Japan for VYVGART for the treatment of ITP expected mid-2023
Efgartigimod Research and Development
argenx aims to solidify its FcRn leadership by expanding the scope of IgG-mediated autoimmune diseases in development and further demonstrating the potential of FcRn blockade in ongoing clinical trials. By the end of 2023, efgartigimod is expected to be approved, in regulatory review or in development in 13 severe autoimmune diseases.
Multiple data readouts expected from ongoing efgartigimod trials in 2023 and 2024:
- ADHERE: Topline data in chronic inflammatory demyelinating polyneuropathy (CIDP) expected in second quarter of 2023
- ADDRESS: Topline data in pemphigus expected in second half of 2023
- ADVANCE-SC: Topline data from SC trial in ITP expected in second half of 2023
- BALLAD and ALKIVIA: Interim data in bullous pemphigoid expected in first half of 2024 and in myositis in second half of 2024
- ALPHA and RHO proof-of-concept (POC) trials through IQVIA collaboration: Topline data from ALPHA in post-COVID-19 postural orthostatic tachycardia syndrome (PC-POTS) expected in fourth quarter of 2023 and from RHO in primary Sjogren’s syndrome expected in 2024
- POC trials underway in membranous nephropathy and lupus nephritis through Zai Lab collaboration
Clinical trials of efgartigimod in additional autoimmune indications to start this year:
- Registrational trial to start in thyroid eye disease in fourth quarter of 2023
- POC trials to start in ANCA-associated vasculitis (ANCA) and antibody mediated rejection (AMR) in kidney transplant in fourth quarter of 2023
Pipeline Progress
argenx is advancing a robust portfolio of innovative clinical programs, including ARGX-117 (C2 inhibitor) and ARGX-119 (muscle-specific kinase (MuSK) agonist). Both programs have the potential to be first-in-class opportunities for multiple severe autoimmune indications.
- ARDA: Interim data from POC trial of ARGX-117 in multifocal motor neuropathy expected mid- 2023
- POC trial of ARGX-117 for prevention of delayed graft function after kidney transplantation expected to start in second half of 2023 following regulatory discussions
- Dermatomyositis selected as third autoimmune indication for development of ARGX-117
- Initiated Phase 1 dose-escalation trial of ARGX-119 in healthy volunteers; subsequent Phase 1b trial to assess early signal detection in patients
Continued investment in Immunology Innovation Program (IIP) to broaden autoimmune pipeline for sustained value creation opportunities
- argenx continues to invest in its discovery engine, the Immunology Innovation Program, to foster a robust innovation ecosystem and drive early-stage pipeline growth. argenx expects to nominate one new development candidate in 2023.
Steve Krognes appointed as non-executive director and Chair of the Audit and Compliance Committee of the Company’s Board of Directors
FOURTH QUARTER AND FULL YEAR 2022 FINANCIAL RESULTS
DETAILS OF THE FINANCIAL RESULTS
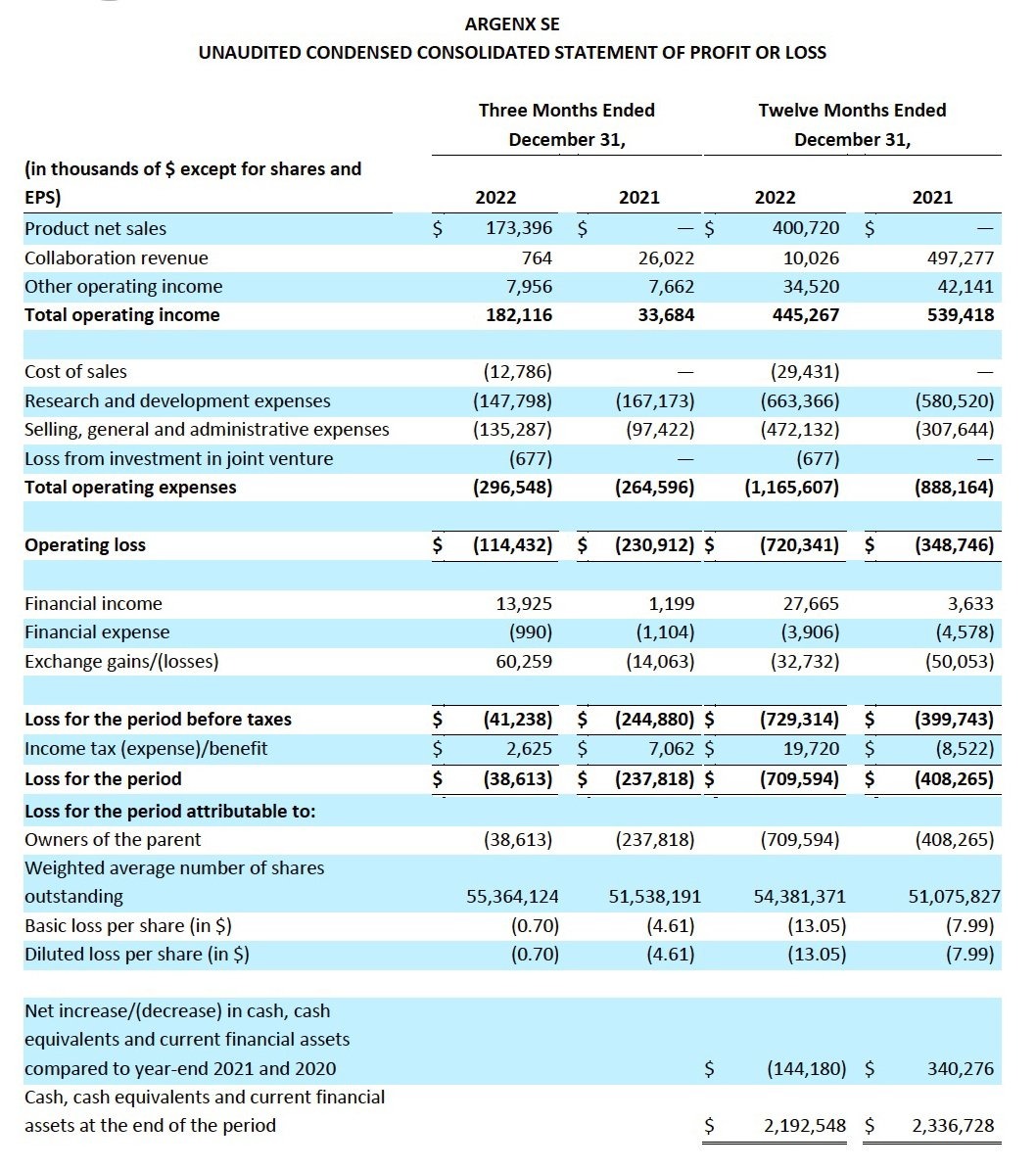
Total operating income for the fourth quarter and year-to-date in 2022 was $182.1 million and $445.3 million, respectively, compared to $33.7 million and $539.4 million for the same periods in 2021, and consists of:
- Product net sales from the sales of VYVGART for the three months ended December 31, 2022 were $173.4 million. The product net sales in the twelve months ended December 31, 2022 were $400.7 million. No product net sales were recognized during the same period in 2021.
- Collaboration revenue for the fourth quarter and year-to-date in 2022 was $0.8 million and
$10 million, respectively, compared to $26 million and $497.3 million for the same periods in 2021. The collaboration revenue during the twelve months ended December 31, 2022 primarily relates to the recognition of milestone revenue following the option exercise by LEO Pharma to enter into a commercial license for ARGX-112. The collaboration revenue for the twelve months ended December 31, 2021 was primarily attributable to the recognition of the transaction price as a consequence of the termination of the collaboration agreement with Janssen, resulting in the recognition of $315.1 million, and the recognition of $177.5 million in collaboration revenue related to the strategic collaboration with Zai Lab, including the development milestone which was triggered by the FDA approval of VYVGART.
- Other operating income for the fourth quarter and year-to-date in 2022 was $7.9 million, and $34.5 million, respectively, compared to $7.7 million and $42.1 million for the same periods in 2021. During the twelve months ended December 31, 2022, and December 31, 2021, the fair value of the argenx profit share in AgomAb Therapeutics NV increased by $4.3 million and $11.2 million respectively. The increase is a result of the extension of a Series B financing round by AgomAb for which argenx maintains a profit share in exchange for granting the license for the use of HGF-mimetic antibodies from the SIMPLE Antibody™ platform.
Total operating expenses for the fourth quarter and year-to-date in 2022 were $296.5 million and $1,165.6 million, respectively, compared to $264.6 million and $888.2 million for the same periods in 2021, and consists of:
- Cost of sales for the fourth quarter and year-to-date in 2022 was $12.8 million and $29.4 million, respectively. The cost of sales was recognized with respect to the sale of VYVGART during 2022. There was no cost of sales recognized in the comparable prior year periods.
- Research and development expenses for the fourth quarter and year-to-date in 2022 were $147.8 million and $663.4 million, respectively, compared to $167.2 million and $580.5 million for the same periods in 2021. The research and development expenses mainly relate to external research and development expenses and personnel expenses incurred in the clinical development of efgartigimod in various indications and the expansion of other clinical and preclinical pipeline candidates. The increase in research and development expense during 2022 is mainly driven by the recognition of the priority review voucher submitted with the BLA filing for SC efgartigimod for the treatment of gMG, which resulted in an expense of $99.1 million.
- Selling, general and administrative expenses for the fourth quarter and year-to-date in 2022 were $135.3 million and $472.1 million, respectively, compared to $97.4 million and $307.6 million for the same periods in 2021. The selling, general and administrative expenses mainly relate to professional and marketing fees linked to the commercialization of VYVGART in the U.S., Japan and the EU and personnel expenses.
- Loss from investment in joint venture for the fourth quarter and year-to-date in 2022 was $0.7 million. The loss recognized was argenx’s share of losses in Oncoverity, Inc. There was no losses from investment in joint venture in the comparable prior year periods.
Financial income for the fourth quarter and year-to-date in 2022 were $13.9 million and $27.7 million respectively, compared to $1.2 million and $3.6 million for the same periods in 2021. The increase in financial income is mainly due to an increase in interest income on current financial assets and cash and cash equivalents attributable to higher interest rates.
Exchange gains/losses for the fourth quarter and year-to-date in 2022 were $60.3 million of gains and $32.7 million of losses, respectively, compared to $14.1 million and $50.1 million of exchange losses for the same periods in 2021. Exchange gains/losses are mainly attributable to unrealized exchange rate losses on the cash, cash equivalents and current financial assets position in Euro.
Income tax for the fourth quarter and year-to-date in 2022 was $2.6 million and $19.7 million of tax benefit, respectively, compared to $7.1 million of tax benefit and $8.5 million of tax expense for the same periods in 2021. Tax benefit for the three months ended December 31, 2022 consists of $12.1 million of income tax expense and $14.7 million of deferred tax income, compared to $1.2 million of income tax expense and $8.2 million of deferred tax income for the same period in 2021.
Net loss for the fourth quarter and year-to-date in 2022 was $38.6 million and $709.6 million, respectively, compared to net loss of $237.8 and $408.3 million for the same periods in 2021.
Cash, cash equivalents and current financial assets totaled $2.2 billion as of December 31, 2022, compared to $2.3 billion as of December 31, 2021. Net change in Cash and cash equivalents and current financial assets is primarily a result of the closing of a global offering of shares, which resulted in the receipt of $761.0 million in net proceeds in March 2022, offset by net cash flows used in operating activities.
FINANCIAL GUIDANCE
Based on current plans to fund anticipated operating expenses, working capital and capital expenditures, argenx expects to utilize up to $500 million cash in 2023.
EXPECTED 2023 FINANCIAL CALENDAR:
- May 4, 2023: Q1 2023 financial results and business update
- July 27, 2023: HY 2023 financial results and business update
- October 27, 2023: Q3 2023 financial results and business update
CONFERENCE CALL DETAILS
The full year 2022 results and fourth quarter business update will be discussed during a conference call and webcast presentation today at 2:30 pm CET/8:30 am ET. A webcast of the live call may be accessed on the Investors section of the argenx website at argenx.com/investors. A replay of the webcast will be available on the argenx website.
Dial-in numbers:
Please dial in 15 minutes prior to the live call.
Belgium | 32 800 50 201 |
France | 33 800 943355 |
Netherlands | 31 20 795 1090 |
United Kingdom | 44 800 358 0970 |
United States | 1 888 415 4250 |
Japan | 81 3 4578 9752 |
Switzerland | 41 43 210 11 32 |
About argenx
argenx is a global immunology company committed to improving the lives of people suffering from severe autoimmune diseases. Partnering with leading academic researchers through its Immunology Innovation Program (IIP), argenx aims to translate immunology breakthroughs into a world-class portfolio of novel antibody-based medicines. argenx developed and is commercializing the first-and- only approved neonatal Fc receptor (FcRn) blocker in the U.S., Japan, and the EU. The Company is evaluating efgartigimod in multiple serious autoimmune diseases and advancing several earlier stage experimental medicines within its therapeutic franchises. For more information, visit www.argenx.com and follow us on LinkedIn, Twitter, and Instagram.
For further information, please contact:
Media:
Erin Murphy
emurphy@argenx.com
Investors:
Beth DelGiacco
bdelgiacco@argenx.com
Forward-looking Statements
The contents of this announcement include statements that are, or may be deemed to be, “forward- looking statements.” These forward-looking statements can be identified by the use of forward- looking terminology, including the terms “believes,” “hope,” “estimates,” “anticipates,” “expects,” “intends,” “may,” “will,” or “should” and include statements argenx makes regarding the impact of the transition of the chief operating officer; its launch strategy to make VYVGART available in the EU, China, Canada and select other regions; the VYVGART multi-dimensional expansion strategy; its expansion through potential regulatory approvals and launches and the planned launch of SC efgartigimod, if approved; the timing of data readouts and new clinical efficacy data; the regulatory reviews and regulatory approval timing in the United States, EU and Japan for SC efgartigimod for the treatment of gMG and the long-term safety and tolerability of SC efgartigimod; the therapeutic potential of its product candidates; the intended results of its strategy and its collaboration partners’, advancement of, and anticipated clinical development and regulatory milestones and plans, including the timing of planned clinical trials; and the design of future clinical trials and the timing and outcome of regulatory filings and regulatory approvals. By their nature, forward-looking statements involve risks and uncertainties, and readers are cautioned that any such forward-looking statements are not guarantees of future performance. argenx’s actual results may differ materially from those predicted by the forward-looking statements as a result of various important factors, including the effects of the COVID-19 pandemic, inflation and deflation and the corresponding fluctuations in interest rates; regional instability and conflicts, such as the conflict between Russia and Ukraine, argenx’s expectations regarding the inherent uncertainties associated with competitive developments, preclinical and clinical trial and product development activities and regulatory approval requirements; argenx’s reliance on collaborations with third parties; estimating the commercial potential of argenx’s product candidates; argenx’s ability to obtain and maintain protection of intellectual property for its technologies and drugs; argenx’s limited operating history; and argenx’s ability to obtain additional funding for operations and to complete the development and commercialization of its product candidates. A further list and description of these risks, uncertainties and other risks can be found in argenx’s U.S. Securities and Exchange Commission (SEC) filings and reports, including in argenx’s most recent annual report on Form 20-F filed with the SEC as well as subsequent filings and reports filed by argenx with the SEC. Given these uncertainties, the reader is advised not to place any undue reliance on such forward-looking statements. These forward-looking statements speak only as of the date of publication of this document. argenx undertakes no obligation to publicly update or revise the information in this press release, including any forward-looking statements, except as may be required by law.